|
Valence electrons control how atoms bond with other atoms to form molecules or crystal structures, and how these bonds break and reform during chemical reactions. 
Parsing the fine points of valence electron motion The results of the pioneering measurements performed at MPQ by the Leone and Krausz groups and their colleagues are reported in the August 5 issue of the journal Nature. Then, with far shorter pulses of extreme ultraviolet light on the 100-attosecond timescale (an attosecond is 10^-18 second, a quintillionth of a second), they were able to precisely measure the effects on the valence electron orbitals. 
The team’s demonstration of attosecond absorption spectroscopy began by first ionizing krypton atoms, removing one or more outer valence electrons with pulses of near-infrared laser light that were typically measured on timescales of a few femtoseconds (a femtosecond is 10^-15 second, a quadrillionth of a second). It’s truly general and can be applied to attosecond electronic dynamics problems in the physics and chemistry of liquids, solids, biological systems, everything.” Leone cites recent work by the Graham Fleming group at Berkeley on the crucial role of coherent dynamics in photosynthesis as an example of its importance, noting that “the method developed by our team for exploring coherent dynamics has never before been available to researchers. “This revealed details of a type of electronic motion – coherent superposition – that can control properties in many systems.” “With a simple system of krypton atoms, we demonstrated, for the first time, that we can measure transient absorption dynamics with attosecond pulses,” says Stephen Leone of Berkeley Lab’s Chemical Sciences Division, who is also a professor of chemistry and physics at UC Berkeley. These oscillations drive electron motion. 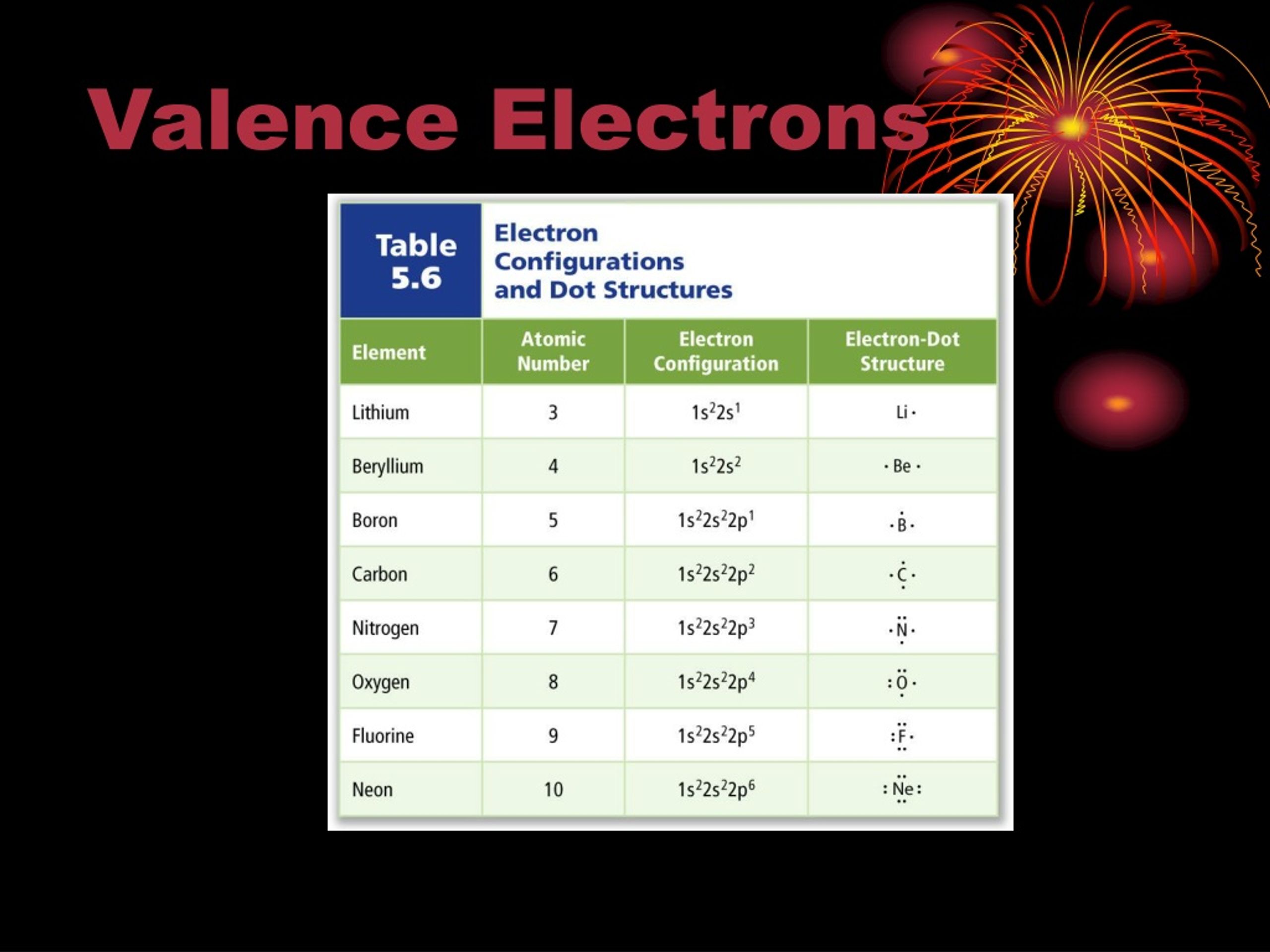
Through a process called attosecond absorption spectroscopy, researchers were able to time the oscillations between simultaneously produced quantum states of valence electrons with great precision. Department of Energy’s Lawrence Berkeley National Laboratory and the University of California at Berkeley has used ultrashort flashes of laser light to directly observe the movement of an atom’s outer electrons for the first time. Researchers have measured oscillations of quantum states (foreground) in the outer orbitals of an ionized krypton atom, oscillations that drive electron motion.Īn international team of scientists led by groups from the Max Planck Institute of Quantum Optics (MPQ) in Garching, Germany, and from the U.S. Berkeley Research Infrastructure Commons (RIC)Ī classical diagram of a krypton atom (background) shows its 36 electrons arranged in shells.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
